Inhibin is a hormone emitted by granulosa cells in the ovarian follicles and by Sertoli cells in the testis. Its physiological role is to stifle secretion of follicle-stimulating hormone (FSH) by the pituitary. They are multifunctional particles engaged with the control of pituitary FSH secretion.
A body of observational and experimental proof from a few species, including the human, supports the concept that its gonadal couriers that exert a physiological negative input control on FSH release at the pituitary gland.
Aside from their fundamental role in the selective control of FSH secretion, its currently accepted as paracrine ovarian and testicular regulators and have multiple paracrine impacts in the uteroplacental unit, representing a favourable marker for male and female infertility, gynaecological and gestational diseases.
Table of Contents
1. Function of Inhibins
In both females and males, it restrains FSH production. It does not inhibit the emission of GnRH from the hypothalamus. However, the overall mechanism varies between the sexes:
a. In Females
- It is made in the gonads, pituitary gland, placenta, corpus luteum and other organs.
- FSH restoratives the secretion of it from the granulosa cells of the ovarian follicles in the ovaries. Thus, it suppresses FSH.
- Inhibin B Reaches a peak in advance- to the mid-follicular stage and a second peak at ovulation.
Inhibin A comes to its peak in the mid-luteal stage. - Its secretion is decreased by GnRH and improved by insulin-like growth factor-1 (IGF-1).
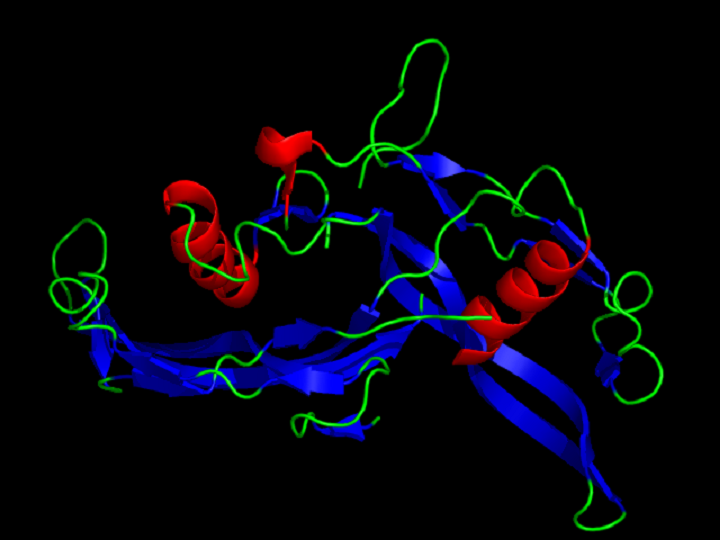
Source: Wikipedia
b. In Males
It is secreted from the Sertoli cells, situated in the seminiferous tubules on the inside the testes. Androgens stimulate its production; this protein may as well help to locally direct spermatogenesis.
c. Activin and Inhibin
Activin and inhibin are two firmly related protein complexes that have straightforwardly inverse biological effects. Distinguished in 1986, activin enhances FSH biosynthesis and secretion, and participates in the regulation of the menstrual cycle. Numerous different functions are exerted by activin, including roles in cell proliferation, differentiation, apoptosis, metabolism, homeostasis, immune response, wound repair, and endocrine activity.
Contrarily, it downregulates FSH synthesis and inhibits FSH secretion. The existence of inhibin was hypothesized as soon as 1916; yet, it was not exhibited to exist till Neena Schwartz and Cornelia Channing’s work in the mid-1970s, after which both proteins were molecularly characterized ten years later.
Activin is a dimer composed of two similar beta subunits. It is additionally a dimer wherein the first component is a beta subunit identical or identical to the beta subunit in activin. However, in contrast to activin, the second element of the inhibin dimer is a more distantly-related alpha subunit.
Activin, inhibin and several other structurally related proteins such as anti-Mullerian hormone, bone morphogenetic protein, and growth differentiation factor belong to the TGF-β protein superfamily. It keeps going as two different isoforms, inhibin A and B. Both isoforms are comprised of 2 subunits, the alpha and beta subunits.
The alpha subunit is alike for both inhibin A and B. However, the beta subunit differs. Inhibin A contains a beta-A subunit, and inhibin B contains a beta-B subunit. Plasma levels of inhibin A and B vary during the menstrual cycle. At menopause, serum inhibin A and B decrease to very low or undetectable levels.
2. Inhibin A
In females, inhibin A is firstly produced by the dominant follicle and corpus luteum: whereas little developing follicles predominantly have inhibin B. Serum inhibin A and B levels vary during the menstrual cycle. At menopause, with the exhaustion of ovarian follicles, serum inhibin A and B reduce to very low or undetectable levels.
Inhibin A is the predominant molecular form of it in maternal flow from 4 weeks of incubation. Even though the precise biological function of inhibin A in pregnancy is obscure, it is evident from recent studies that it could be a better marker of placental function than human chorionic gonadotropin because of its shorter half-life.
Clinical Applications
The possible clinical applications for the measurement of inhibin A in early pregnancy could be in predicting miscarriage, Down’s syndrome, preeclampsia, and fetal growth restriction in the first and second trimester before the onset of the clinical symptoms. The source of its, factors controlling its production, the possible functions of it, and the utilization of its measurement in routine and high-risk pregnancy are evaluated.
The additionally appears to be suitable markers for epithelial tumours of the mucinous type with about 20% of cases having elevated its A levels. In comparison, it is not a very good marker in nonmucinous epithelial tumours. At best, total inhibin is elevated in 15% to 35% of nonmucinous epithelial ovarian cancer growth cases.

3. Inhibin B Female Reproduction
It is a peptide auxin that is initiated firstly by developing antral follicles that are responsive to FSH. As the hair keeps on to grow and become dominant, inhibin B secretion declines. As the growing pool of antral follicles reduces during ageing, levels of inhibin B are diminished.
Studies in ladies have demonstrated that decreased inhibin B secretion is associated with elevated FSH levels and decreased oocyte quality and fertility potential. Thus, It has been utilized as a predictor of ovarian reserve. Yet, the late appearance of inhibin B in the menopausal transition has led to the implication that it may be a superior indicator of ovarian activity than of ovarian reserve, due to its direct connection with growing follicles.
4. Evaluation of Hormonal Status
Measurement
Serum inhibin A and B levels might be measured by specific RIAs, ELISA, or enzyme-linked immunoassay, and reflect the secretion from different inhabitants of granulosa cells. As formerly noted, a characteristic of peptide assays is that different subunits make assays hard.
This is mostly accurate in the case of its assays, which have been highly variable and imprecise. Serum values differ in adult women during the menstrual cycle, with inhibin A being lowest during the follicular phase and peaking during the mid-luteal phase; It exhibits an opposite pattern during fertilization.
Second Trimester
A decreases during the 2nd trimester, whereas It will rise from the 3rd trimester to peak at term. After menopause, levels of the pair its decline markedly. The measurement of it might be helpful in a few conditions. Some granulosa cell tumours produce high amounts of It, which might be useful for postoperative surveillance because it may detect tumour recurrence.
Measurement of inhibin B also has been suggesting for checking certain complications of pregnancy and for evaluating the number of small follicles current in the ovary in the early follicular phase. The measurement may be useful in assessing regenerative ageing because a low level of inhibin B on day 3 may show a declining ovarian reserve.
In men, it is turn out by Sertoli cells and has been examined a useful marker of spermatogenesis. It has as well been used in the differential diagnosis of cryptorchidism versus anorchia.